The five elements miracle soup or broth is commonly used in natural healing for its anti-cancer properties. It was created by Dr. Tateishi Kazu who based the soup on the principles of the five elements theory in Chinese medicine. The harmonizing balance of the forces of yin and yang, acid and alkaline, that generates health; in contrast to the imbalance, which leads to disease.
The five elements are wood, fire, earth, metal and water. Dr. Kazu’s soup is based on the essence of the five elements. He researched, studied and tested over 1,500 types of herbs and plants to come up with the unique healing vegetable soup. Each essence possesses its own color: green, red, yellow, white and black. These relate to the corresponding internal organs: heart, liver, spleen, lung and kidneys. Dr. Kazu used the concept of the five different colors as matched to specific vegetables:
- Daikon Radish Leaves (Green element)
- Daikon Radish (White element)
- Carrots (Red color element)
- Burdock Root (Yellow element)
- Shiitake Mushroom (Black element)
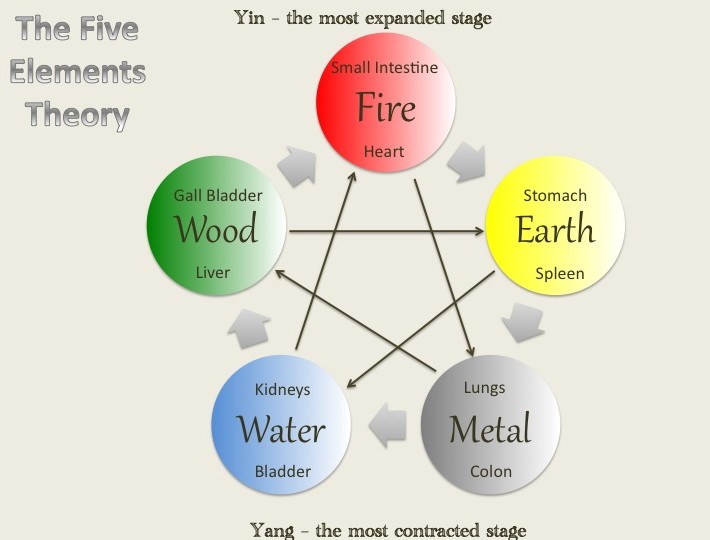
The five elements theory on which Dr. Kazu based his healing soup recipe
This soup is also one of Dr. Baylac’s personal favorite broths for broth fasting. For those of you following a personal detoxification process at home and find yourself in the colder winter climates, utilize this warm broth in your regimen. However, the broth can also be drunk cold for when you are on the go.
This soup can be beneficial for those who are detoxing, fasting, recovering from cancer, and suffering from high blood pressure and arthritis.

5 Elements Soup Recipe
Yield approx.: 3.5 liters, 1 gallon of soup, 14 cups
Ingredients:
- 16 ounces white daikon radish
- 12 ounces white daikon radish greens
- 10 ounces carrot
- 8 ounces burdock root
- 3-5 fresh shitake mushrooms (sundried)
Directions:
- Prepare the vegetables by washing the greens and mushrooms and scrubbing the roots. Do not peel the vegetables. Use them as is for optimal benefit. Chop the vegetables into small pieces.
- There will be no salt or seasonings added to the soup.
- Fill a non-toxic pot with 14 cups of water or three times the quantity of water as the vegetables. Enamel or stainless-steel pots are good choices.
- Add the vegetables and bring to a boil. Once the soup starts to boil, reduce the heat and simmer for two hours.
- Strain the soup to separate the broth. The broth is ready to drink.
- Fill up several 16oz or 8oz mason jars for storage in refrigerator and later use.
- You can also drink the soup simply as a water replacement.
The history and nature of the Miracle Vegetable Soup :
1. The soup was invented in Japan 38 years ago around 1964 by Dean Kazu Tateishi (1934-2001), a physician and a cytology (human cell) expert through experiments with more than 1,500 types of material.
2. We were the only company who had ever met with Dean Kazu Tateishi way from the beginning. Very sad to inform Dean Tateishi passed away in late 2001. He never appointed anyone or any company outside Japan to make the Vegetable Soup for him. However, Dean Tateishi did examine and gave high praise to our soup bag. It was believed that it was the only time that he had seen a soup bag product.
3. Its main function is to strengthen one’s immune system.
4. There are more than 25,000 records of successful examples with the soup in Japan.
5. Celebrities got healed included several Prime Ministers, movie stars, business tycoons in Japan.
6. Ingredients include Turnip, Carrot, and Burdock Root through inter-chemical reactions to provide 30 chemicals to destroy tumor and its abnormal cells.
7. The soup is an ionized solution and helps the body maintain slightly alkaline to give a bad environment for diseases and it results to slow down cancer growing in 4 days and then gradually eliminates the cancer through a course of drinking the soup.
Category: Health & Healing by Tateishi Kazu :
When cancer strikes a family, what course of action should one take? Dr. Tateishi Kazu’s father and brother both died of cancer. And then he found himself suffering from cancer of the duodenum and spreading. His stomach was removed, but the cancer cells had spread to his lungs. He resolved to fight for his life. He researched, studied and tested over 1,500 types of herbs and plants. Eventually he discovered the right combination of ingredients and formulated a unique healing vegetable soup and brown rice tea with its own molecular powers. The vegetables are rich in green chlorophyll, amino acids, iron, phosphorous, calcium, all in a natural form. He based his soup on the principles of Five Elements theory, the harmonizing balance of the forces of yin and yang, acid and alkaline, that engenders health as opposed to the imbalance, which leads to disease. The five elements in our environment are wood, fire, earth, metal and water. Because of the balance of the five elements, the earth is able to produce life. Dr. Tateishi Kazu used the essence of the five elements. Each essence possesses its own color: green, red, yellow, white and black. These relate to the corresponding internal organs: heart, liver, spleen, lung and kidneys. He used the concept of the five different colors as matched to specific vegetables: Green=Daikon Radish Leaves Red=Carrot Yellow=Burdock Root White=Daikon Radish Black=Shiitake Mushrooms This is a remedy for all age groups. When the soup is ingested, it produces thirty different elements for fighting disease. Within three days, it can stop the growth of cancer cells. Even for some last-stage cancer patients, it can lead to 100% remission. Patients who have subsisted on feeding tubes for nourishment, when given the soup, after two days regained their strength. Over 10,000 last-stage cancer patients have used this remedy. 99% are alive and were able to return to work. If you are on medication, consult with a doctor to adjust your dosage. This remedy works for some people. It may not work for you. It is not intended to replace the traditional treatments. What This Remedy May Do:
- Heal Cancer
- Restore injured joints and bone structure
- Slow down aging process
- Rejuvenate the skin
- Heal cataracts
- Heal Liver Disorders
- Lower high blood pressure
- Improve Heart conditions
- Reduce brain tumors and other head injury problems
- Reduce high white blood cell counts
- Improve T-cell function
Vegetable Soup Recipe (2 days quantity)
- 16 ounces White Daikon Radish
- 8-10 ounces White Daikon Radish Greens (Tops)
- 8-10 ounces Carrot
- 8 ounces Burdock Root
- 3-5 fresh Shiitake Mushrooms (sun-dried) If not sun-dried, expose mushrooms to sun again
Directions:
Don’t peel anything! Don’t add any seasoning! Fill a pot with 14 cups of water. When it comes to a boil, reduce the flame and simmer for two hours. Strain and drink. * For last stage cancer patients, take soup as follows: Morning – 150 ml of soup (about 5 ounces) mixed with 30 ml of first morning’s urine (just over an ounce). Do not use the first part of the urine stream because it contains sediment. Only use your own urine. Urine is not a toxin. It is a medically proven, sterile, highly concentrated solution of excess nutrients, vitamins, hormones, enzymes and critical antibodies that cannot be duplicated. Stop adding the urine after three months. Afternoon – 200 ml of soup (about 7 ounces). Evening – 200 ml of soup. Note: when using urine therapy with the soup you will periodically feel discomfort in the diseased area once the healing process begins and the body starts to reverse the damage.
Brown Rice Tea Recipe Another remedy that can be used in conjunction with the vegetable soup is brown rice tea. It is a good diuretic for ridding excess water from the body. For diabetics, it can bring the sugar level down by producing more natural insulin in the body. It cleanses the blood and blood vessels. A person with a heart condition drinking both soups in 20 days will be able to regulate their problem by drinking three cups per day. For a regular cancer patient, about two cups. 1 cup brown rice 16 cups water Note: only cook in stainless steel or glass. Do not mix with any other ingredients. Don’t be “creative.” Directions: 1. Roast the rice without oil until dark brown but don’t burn. Boil 8 cups of water and pour roasted rice into water. Turn off flame, cover and let sit five minutes. Strain out rice and reserve liquid. 2. Boil 8 more cups of water. Put the remaining rice back in the water. Cover, lower flame and simmer for five minutes. Strain rice out. Mix the two rice waters together. ——————————————————————————–
Special Notes: The amount one needs depends on the condition. The more serious the condition, the more tea.
1. Do not drink tea while taking a high protein substance.
2. Don’t drink the vegetable soup and brown rice tea together. Wait at least 15 minutes apart.
3. If you use this remedy regularly you will never be sick.
——————————————————————————–
For high blood pressure, drink the two soups alternately but slowly. Blood pressure will come down after one month. Talk to a doctor about reducing medication, but don’t stop medication right away. Use remedy for minimum of one month.
Progress of Healing After three days of use, cancer cells stop growing. After a minimum of one-month normal cells begin to grow back.